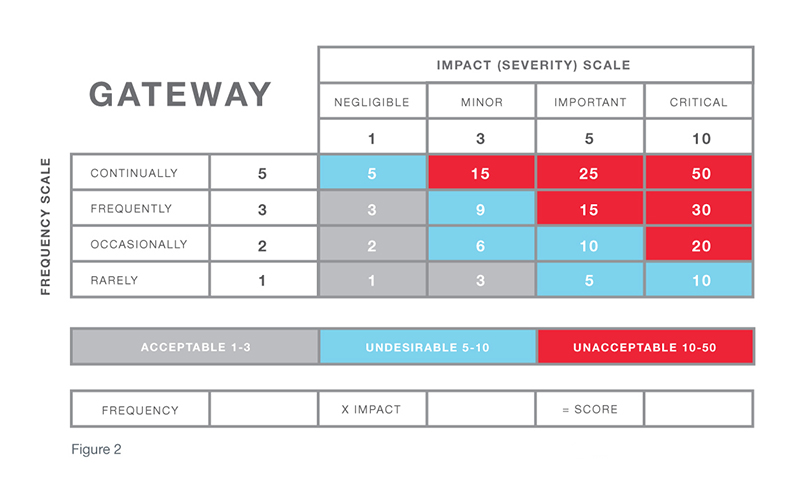
Corrective and Preventive Action (CAPA) Process Planning for Clinical Research Professionals — Clinical Pathways

Principles of corrective Action and Preventive Action :CAPA: A Handbook for Quality Professionals in Medical device and Pharmaceutical Industries: MUCHEMU PhD, Dr DAVID NAKASALA: 9781790621101: Amazon.com: Books